Physical Mixture of Gold Nanoparticles and Carbon Dots Enable Sensing of Cyanide Ions in Water in Dual Modes
Changerath R, Kunnatheery S and Roy Joseph
Changerath R1*, Kunnatheery S1 and Dr Roy Joseph2
1Laboratory for Polymer Analysis, Biomedical Technology Wing, Sree Chitra Tirunal Institute for Medical Sciences & Technology, Thiruvananthapuram, Kerala, India
2Scientist 'G' at Sree Chitra Tirunal Institute for Medical Sciences and Technology, Trivandrum, India
- *Corresponding Author:
- Changerath R
Laboratory for Polymer Analysis
Biomedical Technology Wing
Sree Chitra Tirunal Institute for Medical Sciences & Technology
Thiruvananthapuram, Kerala, India
Tel: 091- 471 -2520268
E-mail: radhakumary@sctimst.ac.in
Received Date: June 14, 2019; Accepted Date: June 28, 2019; Published Date: July 02, 2019
Citation: Changerath R, Kunnatheery S, Joseph R (2019) Physical Mixture of Gold Nanoparticles and Carbon Dots Enable Sensing of Cyanide Ions in Water in Dual Modes. Nano Res Appl Vol.5 No.1:3. DOI: 10.36648/2471-9838.5.1.40
Abstract
A novel, cost effective, sensitive and environmentally friendly approach using a physical mixture of carbon dots (CDs) and gold nanoparticles (AuNPs) is reported for the detection of cyanide ions (CN-) in water. We observed significant quenching of fluorescence of CDs by AuNPs and the restoration of fluorescence by the removal of AuNPs. A dual mode sensing approach for the estimation of CN- in water is carved out of this observation exploiting both the FRET between CDs and AuNPs and the well-known gold leaching by CN-. This technique does not involve any complex chemistry or any tedious synthetic routes and can be used for the sensing of cyanide in aqueous media. The method is sensitive up to a concentration of 1 mgL-1 (1 μg mL-1) of CN- in water and its presence/absence can also be visualized by naked eye by illuminating the solution using light at 365 nm.
Keywords
Carbon dot; Gold nanoparticle; Fluorescence quenching; Cyanide ion
Introduction
Through ages, cyanide is the most feared anions in the environment. The lethality and its action on living systems are well documented. CN- causes death of living organisms even at lower concentrations unlike heavy metal ions which affect health only at later stage by accumulating in the body. Though CN- is extremely toxic, it is widely used throughout the world in various sectors of industry. Accidental CN- release in wastewater or rivers may lead serious contamination of ground water and even drinking water [1]. The world health organization fixed the maximum acceptable level of cyanide in drinking water at 1.9 μM1 and the United States Environmental Protection Agency (EPA) has set the maximum contaminant level (MCL) for cyanide in drinking water at 0.2 mgL. The extreme toxicity of cyanide in physiological systems, as well as the continuing environmental concern caused by its widespread industrial use has led to the development of varied analytical approaches for its detection [2].
Several methods are reported in the literature using fluorimetry, spectrophotometry, chromatography, titrimetry, electrochemical devices and chemodosimetery for the determination of cyanide [3-19]. Numerous studies on AuNP based colorimetric sensing of cyanide ions are also reported [20-23]. Most of these methods need tedious synthetic steps, use of organic solvents, sophisticated instruments and are not suitable for on-site cyanide monitoring.
Semiconductor quantum dots (QDs) have become one of the heavily studied materials in diverse fields due to its high emission quantum yields and size tunable emission profiles. Their serious toxicity even at relatively low concentrations and limited aqueous solubility restrict their applications particularly in bio and environmental domains [24]. In comparison to the well-established semiconductor QDs, carbon quantum dots (CDs) have attracted tremendous attention because of their interesting properties, such as non-blinking, water solubility, and nontoxicity [24]. AuNPs are well known as quenchers. This property, in fact, has been used in designing novel sensors employing fluorophores such as semiconductor QDs. Fluorescence Resonance Energy Transfer (FRET) between QDs and AuNPs are used for the assay of biomolecules that inhibit the specific interactions between biomolecules [25]. Quenching of fluorescence of CDs by heavy metal ions have been explored and subsequently used as detection methods for cations. Recently significant efforts related to the construction of sensors based on CDs have been emerged [26]. We observed the quenching of fluorescence of CDs by AuNPs.
We reasoned that quenched fluorescence could be revived by the simple removal of AuNPs. Simultaneously analyte induced leaching of Au can also be followed by the reduction in SPR peak of AuNPs. Reliability and applications of an analytical approach would be better and wider if the measurement can be performed in more than one mode. Present communication reports for the first time a one-shot method using unmodified CDs and AuNPs for the detection and estimation of cyanide ions in water in dual modes by monitoring fluorescence and optical absorbance.
Experimental Procedure
Materials
Chloroauric acid (HAuCl4.3H2O), Trisodium citrate, Citric acid anhydrate, PEG diamine and Rhodamine B were purchased from Sigma-Aldrich, Bangalore, India. Potassium cyanide was a gift from the toxicology laboratory, Biomedical Technology Wing, SCTIMST. All other chemicals used were of analytical grade from Merck India Ltd, Mumbai, India.
Preparation of gold nanoparticles
AuNPs were synthesized as reported by Turkevich et al. [27]. Briefly, to a boiling solution of 20 mL of 1.0 mM HAuCl4, 2 mL of a 1% solution of trisodium citrate dihydrate was added under constant stirring. The contents were removed from the hot plate when the solution turned red. The cooled contents were kept under refrigeration until its usage.
Synthesis of Carbon Dots (CDs)
Carbon dots were synthesized as reported by Goh et al. [28]. Briefly, 9 ml of glycerin and 600 mg of PEG diamine (MW ~1800) were taken in a 100 mL three-neck flask degassed with nitrogen. When the temperature was raised to 250°C, 600 mg of citric acid was added and allowed to react at this temperature for 3hrs. The resulting product was cooled to room temperature and dialyzed against distilled water using a cellulose ester dialysis membrane [molecular weight cut off 3500) for 2 days in order to remove any unreacted reactants. The purified CD was freeze dried and kept refrigerated until use.
Instrumentation
The UV-Visible absorption spectra of the Au and CD-Au were recorded using a UV-Visible spectrophotometer, Varian, Cary 100 Bio, Melbourne, Australia and the fluorescence intensity of the as prepared CD and CD-Au were measured using a Spectrofluorometer, Varian, Cary Eclipse model EL 0507, Melbourne, Australia. Fourier Transform Infra-Red (FTIR) spectrum of CD was recorded in the range 600-4000 cm-1 on a Nicolet 5700 FTIR Spectrometer, Nicolet Inc, Madison, USA using a Diamond ATR accessory. The technique of Dynamic Light Scattering (DLS), Malvern Instruments Ltd, UK was used for the determination of the zeta potential of the CD, CD-Au and CD-Au- CN. High Resolution Transmission electron microscopy (HRTEM) was performed in a FEI, TECNAI S Twin microscopy with an accelerating voltage of 100 KV. The sample solutions (CD) were prepared by dispersion under an ultrasonic vibrator. They were then deposited on a formvar coated copper grid and dried in a vacuum at room temperature before observation. Transmission Electron Microscopic (TEM) images were obtained on a Hitachi, H 7650 microscope, Hitachi, Tokyo, Japan. The colloidal solution was deposited onto a 200-mesh copper grid coated with a formvar film and dried overnight.
Measurement of fluorescent quantum yields of CDs
The quantum yield (QY) of CDs was determined using a comparative method. The method involves multiple references with known QYs and provides much high accuracy by calculating the slope of the line generated by plotting the integrated fluorescence intensity against the absorbance for multiple concentrations of fluorophore. Rhodamine B in water (QY = 31%) was used as the standard to calculate the QY of the CDs. It is reported that the solution absorbance at the excitation wavelength for either the sample or the standard should not exceed 0.1 to guard against any significant losses due to inner filtration effects [29]. Hence absorbance values chosen were 0.008, 0.03, 0.05 and 0.1. Emission spectra of the solutions were recorded using a spectrofluorometer at an excitation wavelength of 350 nm. Integrated fluorescence intensity was calculated which is the area under the curve of the emission spectra. A graph was plotted with the integrated fluorescence intensity against the absorbance and linear fit curves were generated from which we obtain the slope values. Calculation was done based on the following equation,
Q = QR (m/mR) (n2/nR2) --------- (1)
Where m is the slope of the line, n is the refractive index of the solvent, subscript R refers to the quantum yield of the standard [30]. Here for both the standard and the sample solvent is water; hence n2/nR2 is 1.
Detection of cyanide in water samples
Aqueous solution of potassium cyanide (0.1 M) was prepared and diluted 1:10 to get 0.01 M. 1 mL of CD solution (0.1 mg/ mL) was quenched with gold nanoparticles (AuNPs) (0.1 mg/ mL) and this CD-Au mixture is used for the detection of CN-. Different amounts of CN- ranging from 5 mgL-1 (μgmL-1) to 180 mgL-1 were added to the fluorescence quenched CD-Au and measured both the decrease in the SPR absorption intensity of AuNPs and the increase in fluorescence emission intensity of the CDs. Dilution factor has been taken into account in fixing the intensity of absorption/emission peaks. To confirm the precision and recovery of the probe each set of experiment was carried out in triplicate and similar results within the maximum error of 2-3% were obtained.
Results and Discussion
We synthesized CDs as reported elsewhere [28]. The quantum yield of the CDs was determined to be 19.3%. FTIR spectrum, depicted in Figure S1, shows characteristic peaks of CDs as reported by Goh et al. along with the strong C-H stretching vibrations of PEG at 2869 cm-1 [28]. The fluorescence of CDs (1 mL, 0.1 mg/mL) is quenched with 1 mL AuNP (0.1 mg/mL) and the obtained mixture is named as CD-Au. The interaction of CD with AuNPs does not cause any change in the optical properties of the AuNPs as evident from absorption spectrum shown in Figure S2. It is also clear that the CDs does not induce any aggregation of the AuNPs since the SPR maximum of the AuNPs is not red shifted in the CD-Au and no additional peak is observed beyond the SPR peak. Aggregated structures would have shown a color change or additional peak. The decrease in intensity of the SPR absorption of AuNPs is due to the dilution effect caused by the addition of 1 mL of CD.
The HRTEM micrograph indicates that as prepared CD has an average size of 3-5 nm which agrees with the earlier literature (Figure 1A). The size of the particles of CD-Au is ~14 nm as shown in TEM micrograph (Figure 1B). It appears that the spherical structures of bigger sizes are that of AuNPs. TEM image of CD-Au on interaction with CN- is shown in Figure 1C. Most of the AuNPs [spherical particles in Figure 1B)] are leached away by CNas reflected by the image. Zeta potential of CD is -20.9 mV and it is reduced to -3.4 mV for CD-Au. It is reasoned that the reduction is due to the interaction of CD and AuNPs. This feasibility, in fact, is well reflected in the increase in Zeta potential to -36 mV on interaction with CN- which is known to remove Au particles (Table 1).
| Sample code | Zeta Potential (mV) |
|---|---|
| CD | -20.9 ± 4.7 |
| CD-Au | -3.4 ± 4.6 |
| CD-Au-CN | -36.2 ± 6.4 |
Table 1: Change in Zeta potential of CDs when mixed with AuNPs and CN ions.
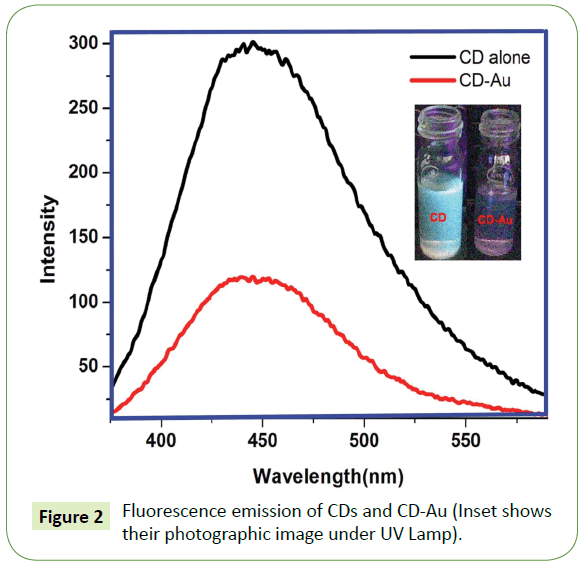
It is the Fluorescence spectra of as prepared CDs and CD-Au (Figure 2). It can be seen that ~62% quenching of the fluorescence emission of CDs were affected by AuNPs. Complete quenching of fluorescence cannot be expected since the AuNPs are randomly distributed in the medium. Photographic image of CD and CD-Au under UV lamp shown in the inset of Figure 1 clearly reflect the quenching of CD by AuNPs. The image is predominately red, characteristic color of AuNPs. On adding CN- to CD-Au mixture, AuNPs are quantitatively removed and simultaneously fluorescence intensity of the CDs is reinstated thus by enabling estimation of CN- ions in aqueous samples in dual modes.
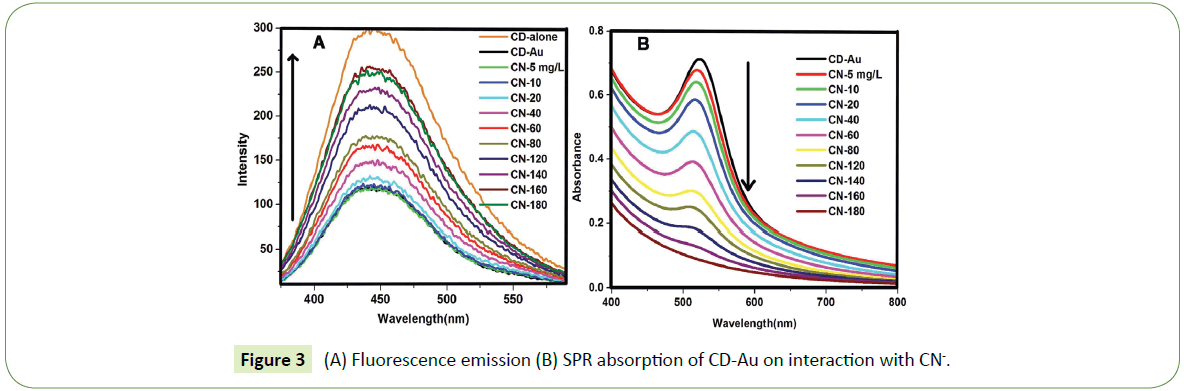
The photographic images under UV lamp depicted in Figure S3 A-E show the quenching of the fluorescence of CDs by AuNPs and the images in Figure S3 F-J show the restoration of the fluorescence on adding CN-. Additionally, Figures S4 A and B is the photographic images of CD-Au and CD-Au-CN under visible light. It can be seen that CN- completely leaches away the AuNPs (the red color is faded in B) further supporting the TEM images in Figure S3 C. Figures 3A and 3B depicts the increase in intensity of fluorescence emission of CD-Au and the decrease in absorbance of the SPR of the AuNPs on adding CN-. Fluorescence intensity is revived proportionally with concomitant reduction in the SPR absorbance.
Interestingly the increase in fluorescence and the reduction in absorbance are found to be linear with respect to the concentration of CN- ions (Figures S5 A and B). This method can be used to detect CN- concentration as low as 1 mg/L (1 μg/mL) in water and its presence/absence can also be visualized by naked eye by illuminating the solution using light at 365 nm.
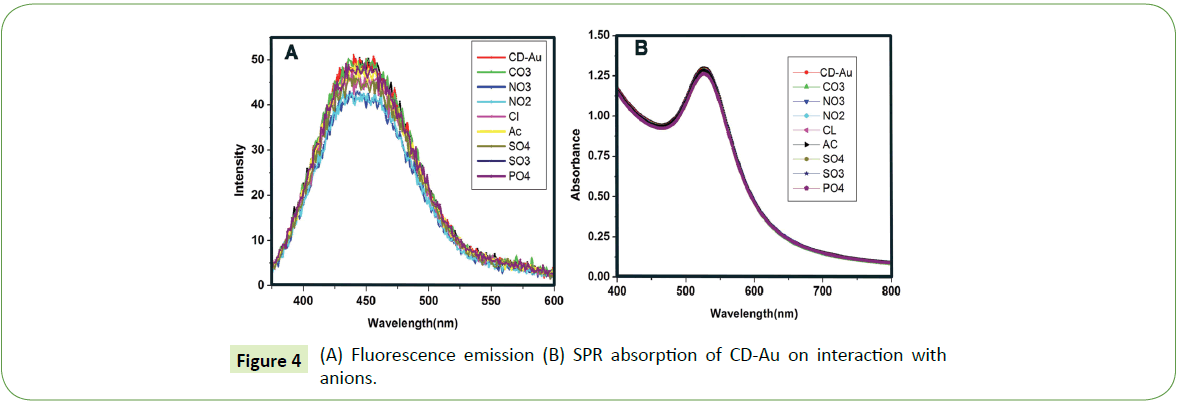
We found that commonly occurring anions (1000 mgL-1) neither enhanced the fluorescence nor reduced the SPR intensity as shown in Figure 4 apparently suggesting that common anions didn’t have any adverse influence on the detection of CN ions. Several authors have reported such chromofluorogenic detection methods for the sensing of CN ions. However, a dual sensing method for the CN ion using the same probe is not found yet [31- 33]. Yadav and Singh have reported a dual anion sensing method for arsenite and cyanide ions [34].
We used this method for checking cyanide contamination in water samples collected from our campus and also outside the campus. None of the samples showed any detectable quantity of CN- However, water spiked with 100 mgL-1 showed the restoration of fluorescence intensity and the corresponding decrease in the SPR absorbance of the CD-Au as depicted in Figure S6.
Earlier reports have shown that fluorescence of many fluorophores including conjugated polymers can be quenched by the addition of trace amounts of AuNPs. These studies indicate that the mechanism of quenching is predominately by energy transfer (FRET). Though we have not investigated the possible mechanism of quenching of fluorescence of CDs by AuNPs, we reasoned that FRET is operative here considering the fact that there is significant spectral overlap of the fluorescence of CDs and the absorption spectra of AuNPs.
Conclusion
In conclusion, a dual sensing approach for the estimation of CNin water is reported for the first time exploiting both the FRET between CDs and AuNPs and the well-known gold leaching by CN-. The method does not involve any complex chemistry or any tedious synthetic routes and can be used for the sensing of cyanide presence in aqueous media. The method is sensitive up to a concentration of 1 mg/L (1 μgmL-1) of CN- in water and hence can be used for the preliminary checking of cyanide contamination in drinking water. The method seems to have wider prospects since the analysis can be performed in dual modes.
Acknowledgments
The authors want to thank the Department of Biotechnology, New Delhi for financial support. We are also thankful to Ms. S. Many and Mr. W. Paul for their technical assistance in getting the TEM images and DLS data.
References
- Shan D, Mousty C, Cosiner S (2003) Layered double hydroxides: An attractive material for electrochemical biosensor. Design. Anal. Chem 75: 3872-3879.
- Xu Z, Chen X, Kim H, Yoon J (2010) Sensors for the optical detection of cyanide ion. Chem Soc Rev 39: 127-137.
- Suzuki T, Hiolki A, Kurahashi M (2003) Development of a method for estimating an accurate equivalence point in nickel titration of cyanide ions. Anal Chim Acta 476: 159-165.
- Safavi A, Maleki N, Shahbaazi H (2004) Indirect determination of cyanide ion and hydrogen cyanide by adsorptive stripping voltammetry at a mercury electrode. Anal Chim Acta 503: 213-221.
- Ma J, Dasgupta P (2010) Recent developments in cyanide detection: A review. Anal Chim Acta 673: 117-125.
- Suresh K, Rao K, Nair T (1997) Genetic variability in photosynthetic rate and leaf characters in Brassicaceae coenospecies. Photosynthetica 33: 173-178.
- Christison T, Rohrer J (2007) Direct determination of free cyanide in drinking water by ion chromatography with pulsed amperometric detection. J Chromatogr A 1155: 31-39.
- Lee H, Chung Y, Ahn K (2008) Selective fluorescence sensing of cyanide with an o-(carboxamido) trifluoroacetophenone fused with a cyano-1,2-diphenylethylene fluorophore. Tetrahedron Letters49: 5544-5547.
- Badugua R, Lakowicza J, Geddes J (2005) Cyanide-sensitive fluorescent probes. Dyes and Pigments64: 49-55.
- Badugua R, Lakowicza J, Geddes C (2004) Excitation and emission wavelength ratiometric cyanide-sensitive probes for physiological sensing. Analytical Biochemistry 327: 82-90.
- Badugua R, Lakowicza J, Geddes C (2005) Enhanced fluorescence cyanide detection at physiologically lethal levels: Reduced ICT-Based signal transduction. J Am Chem Soc 127: 3635-3641.
- Lee K , Huh J, Kim T, Do Y, Lee M (2008) Electron-beam-induced transition aluminas from aluminum trihydroxide. Scriptamat 59: 1022-1025.
- Gimeno N, Li X, Durrant J, Vilar R (2008) Cyanide sensing with organic dyes: Studies in solution and on nanostructured Al2O3 Surfaces. Chem. Eur J 14: 3006-3012.
- Legako J, White B, Harmon H (2003) Rapid reagent-less detection of competitive inhibitors of butyrylcholinesterase. Sensors and Actuators B: Chemical 91: 138-142.
- Gee H, Lee C, Jeong Y, Woo-Dong J (2011) Highly sensitive and selective cyanide detection via Cu2+ complex ligand exchange. Chem. Commun 47: 11963-11965.
- Chung S, Nam S , Lim J, Park S, Yoon J (2009) A highly selective cyanide sensing in water via fluorescence change and its application to in vivo imaging. Chem Commun (Camb) 28: 2866-2868.
- Ajayakumar M, Mukhopadhyay P, Yadav S, Ghosh S (2010) Single-electron transfer driven cyanide sensing: A new multimodal approach. Org Lett 12: 2646-2649.
- Kim H , Lee H, Lee J, Choi D, Jung J, et al. (2011) Bisindole anchored mesoporous silica nanoparticles for cyanide sensing in aqueous media. Chem Commun 47: 10918-10920.
- Mashraqui S, Betkar R, Chandiramani M, Estarellas C, Frontera A (2011) Design of a dual sensing highly selective cyanide chemodosimeter based on pyridinium ring chemistry. New J Chem 35: 57-60.
- Senapati D, Dasary S, Singh A, Senapati T, Yu H, et al. (2011) A labelÃÆâÃâââ¬ÃâÃÂfree goldÃÆâÃâââ¬ÃâÃÂnanoparticleÃÆâÃâââ¬ÃâÃÂbased sers assay for direct cyanide detection at the partsÃÆâÃâââ¬ÃâÃÂperÃÆâÃâââ¬ÃâÃÂtrillion level. Chem Eur J17: 8445-8451.
- Liu C, Tseng W (2011) Colorimetric assay for cyanide and cyanogenic glycoside using polysorbate 40-stabilized gold nanoparticles. Chem Commun 47: 2550-2552.
- Kim M, Kim S, Jang H, Yi S, Seo S, et al. (2010) A gold nanoparticle-based colorimetric sensing ensemble for the colorimetric detection of cyanide ions in aqueous solution. Tetrahedron Letters 51: 4712-4716.
- Zhang Z, Zhang J, Qu C, Pan D, Chen Z, et al. (2012) Label free colorimetric sensing of thiocyanate based on inducing aggregation of Tween 20-stabilized gold nanoparticles. Analyst 137: 2682-2686.
- Zhou L, Lin Y, Huang Z, Ren J, Qu X (2012) Carbon nanodots as fluorescence probe for rapid, sensitive, and label-free detection of Hg2+ and biothiols in complex matrices. Chem Commun 48: 1147-1149.
- Eunkeu O, Mi-Young H, Dohoon L, Sung-Hun N, Hyun C, et al. (2005) Inhibition assay of biomolecules based on fluorescence resonance energy transfer (FRET) between quantum dots and gold nanoparticles. J Am Chem Soc127: 3270-3271.
- Li H, Kang Z, Liu Y, Lee S (2012) Carbon nanodots: Synthesis, properties and applications. J Mater Chem 22: 24230-24253.
- Turkevich J, Stevenson P, Hillier J (1951) A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc 11: 55-75.
- Goh E, Kim K, Kim Y, Jung H, Beak S, et al. (2012) Bioimaging of hyaluronic acid derivatives using nanosized carbon dots. Biomacromolecules 13: 2554-2561.
- Zhai X, Zhang P, Liu C, Bai T, Li W, et al. (2012) Highly luminescent carbon nanodots by microwave-assisted pyrolysis. Chem. Commun 48: 7955-7957.
- Williams A, Winfield S, Miller J (1983) Relative fluorescence quantum yields using a computer-controlled luminescence spectrometer. Analyst 108: 1067-1070.
- Kim J, Ramalingam M, Son Y (2017) A reaction based colorimetric chemosensor for the detection of cyanide ion in aqueous solution. Sensor Actuat B: Chem 246: 319-326.
- Abdinejab T, Zamanloo M, Mahmoodi N, Alizadeh T, Shamkhali A (2019) Colorimetric sensing of cyanide ion by pyromellitic diimides synthesized in one step from commercially available reactants. J Photochem 371: 17-24.
- Alizadeh A, Ghouzivand S, Khodai M, Ardalani M (2016) An interesting spectroscopic method for chromofluorogenic detection of cyanide ion in aqueous solution: Disruption of intramolecular charge transfer (ICT). J Chem Sci 128: 537-543.
- Yadav N, Singh A (2016) Dual anion colorimetric and fluorometric sensing of arsenite and cyanide ions. RSC Adv 6: 100136-100144.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences